Brilliance in diamond dust
Post Date: 17 Mar 2014 Viewed: 338
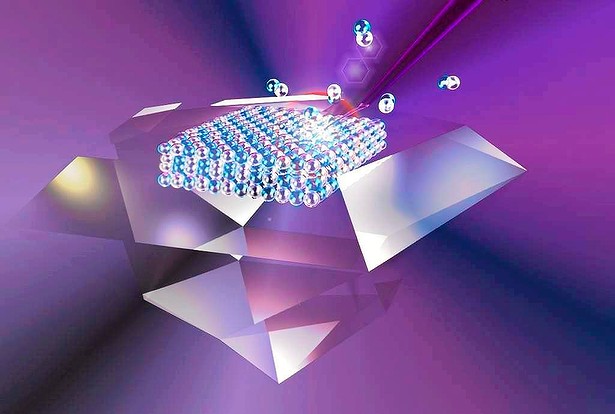
DIAMONDS TO DUST
One aim of future research is to ultimately confine the light interaction to the atomic scale and to demonstrate selective single atom removal.
Small, it seems, is never quite small enough. In their relentless quest to build ever-more minuscule and compact electronic devices, scientists have attempted to manipulate a variety of materials down to the atomic level.
For many reasons, this has proved tough to achieve. Now, a team of Australian researchers has succeeded in using intense pulses of laser light to move individual atoms in substances as rock-solid as diamonds.
The breakthrough is likely to lead to new types of nano-scale devices measuring just billionths of a metre, including minute sensors, super-small and fast electronic components and data storage systems, quantum computers and perhaps a new generation of high-powered lasers on tiny chips.
The discovery, reported in the British journal Nature Communications, resulted more from serendipity than planning. "To our surprise, we found that ultraviolet lasers could be used to target specific atoms," says team leader Richard Mildren, of Macquarie University in Sydney.
"We knew that UV lasers could eject atoms from the surface of diamonds – even at very low light levels," Associate Professor Mildren explains. "But there was no clue to suggest that this process could be harnessed to remove a single targeted atom."
The telling clue, he says, came from ongoing research using an intense UV laser to slice through small sections of diamond.
Diamonds derive their hardness from the way their carbon atoms are arranged in an extremely rigid grid, known as a crystal lattice. The rigidity results from each atom being bound tightly to four other carbon atoms.
Although diamonds are generally transparent to UV rays, a sliver of the light is absorbed very close to the surface. "We think it may occur in the top one or two rows of atoms," Professor Mildren says. "The surface of diamond is normally covered in oxygen atoms and we suspect the carbon is released in the form of carbon monoxide molecules."
The added energy, he says, is enough to break the chemical bonds that normally bind carbon atoms to the surface.
The scientists found that it takes the energy of two UV light particles, or photons, to dislodge one carbon atom. "Carbon atoms are ejected from the surface one by one," he says. "The rate at which this happens is very predictable."
Exactly how the energy is absorbed, and leads to the bonds being broken, is not yet well understood. "This is something we need to work on."
Shedding light
Not any old light does the trick. The diamonds his team worked on were exposed to a very specific form of light pulses in the UV-C band. These are the sun's harshest rays that are largely filtered out by Earth's ozone layer.
A few seconds after being bombarded with light pulses, the diamonds developed small pits on their surface. "The rate of mass loss in the diamond fell notably for lower light levels," Professor Mildren says. "But the etching process still continued – albeit at a slower and slower pace."
The rate of this etching is so slow that it is not noticeable under normal circumstances. In fact, even under very bright conditions, such as intense sunlight or a UV tanning lamp, it would take roughly the age of the universe – almost 14 billion years – to make an appreciable impact on a diamond.
This is where lasers come in handy. These are basically devices that emit intense beams of light by amplifying the light waves using a process called stimulated emission of electromagnetic radiation. The term laser, in fact, is an acronym for "light amplification by stimulated emission of radiation".
Beams emitted in this way differ from other sources of light because they emit the light coherently. In essence, this means that a "hot" beam can be intensified and concentrated onto a very small area. This allows them to cut or weld through virtually any solid material.
The laser's ability to cut out components on dimensions much smaller than the width of a human hair makes them prized tools in high-tech industries, including electronics and car making.
But at smaller scales – such as the distances between atoms – lasers were, until recently, generally considered to be quite ineffectual.
The problem, Professor Mildren says, is that laser cutting and material processing have relied on the heat produced by a laser's beam, in many cases stripping electrons from their parent atomic nuclei. The smallest cuts that could be made depended on the amount of heat transferred to the surface.
"There are now promising signs that it is possible to use lasers to carve up a material with atomic resolution – that is to pick apart a substance atom by atom by using a light beam to snip the chemical bonds holding the individual atoms together," he explains.
Key result
The researchers have experimented with their lasers, for example machining a variety of surfaces.
Examination of the machined surfaces using a high-powered electron microscope showed the formation of a curious pattern of regular nano-structures, Professor Mildren says.
"The key observation came when varying the light beam's polarisation – that is, the direction of the light wave's oscillating movement. The particular shape and orientation of these patterns altered with the way chemical bonds of surface atoms lined up with the polarisation."
This surprising observation, he says, provided the essential clue that the light was somehow interacting with individual bonds. "It also showed that chemical bonds can be broken before there is any significant dissipation of energy to cause damage to the surrounding area."
Applications
Low-cost production of high-quality diamonds from synthetic sources is driving developments in areas such as ultra-fast electronics, quantum computing devices and miniature high-powered diamond lasers.
"Having a new tool to construct and manipulate diamond devices at the ultimate level of resolution is very exciting for developing these future technologies," Professor Mildren says. "We have already shown that it's possible to make diamond structures of less than 20 nanometres – within the size range of large molecules. This is many tens of times smaller than what could previously be achieved, and suitably small to be of immediate use in applications such as super-low friction surfaces and advanced light sources."
The next goal, he says, is to develop ways to treat single or small groups of atoms. "We would like to manipulate surfaces with single-atom precision, or more than 10,000 times smaller than that possible by standard laser machining techniques. This is an area full of interesting challenges for confining a laser beam sufficiently to gain the necessary level of control."
Professor Mildren and colleagues Andrew Lehmann and Carlo Bradac admit the mechanisms behind this process are not yet well understood. "So it is important to study the process in greater detail, asking such questions as: how is the light absorbed? And: how are the chemical bonds broken without significant leakage of energy into surrounding areas?"
That this effect was first detected in diamonds is no coincidence, Professor Mildren says. "Although they have been known for thousands of years, diamonds are only now gaining true importance in science and technology. They have very highly defined bonds that are relatively disconnected from neighbouring atoms. So another key question is this: how many materials other than diamond can we laser-pick apart like this? And what might be the consequences?"



